The Hand Warmer Project
Objective
to design and create a hand warmer using chemistry to make an effective, safe, environmentally friendly, and inexpensive hand warmer, so it can be sold in the San Marin Student Store.
to design and create a hand warmer using chemistry to make an effective, safe, environmentally friendly, and inexpensive hand warmer, so it can be sold in the San Marin Student Store.
Process
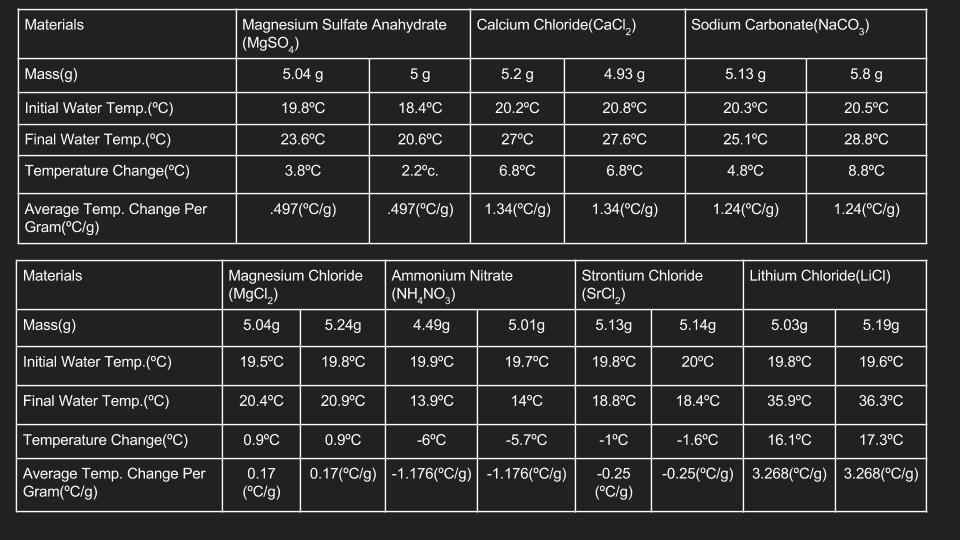
In this project, I was grouped with Jason, Kyle, and gabby. Our design process went through many different steps. We started of knowing one major fact: we needed to use an exothermic reaction to propel our hand warmer, since exothermic reactions expel heat. We then began by testing ionic salts in water. The data for this is below.
In this project, I was grouped with Jason, Kyle, and gabby. Our design process went through many different steps. We started of knowing one major fact: we needed to use an exothermic reaction to propel our hand warmer, since exothermic reactions expel heat. We then began by testing ionic salts in water. The data for this is below.
The tests showed that when mixed with water, lithium chloride produces the most heat. From there, we then tested different models of handwarmers. After 5 different prototypes, we eventually came up with this finalized design.

Specs
- Is 1.5x3.5 inches
- Made of 3 different parts
- A 1x1 inch bag full of water beads (the green)
- A 1.25x3.25 plastic bag that encases the lithium chloride and the bag of water beads
- A 1.5x3.5 inch cotton bag that encases the plastic bag
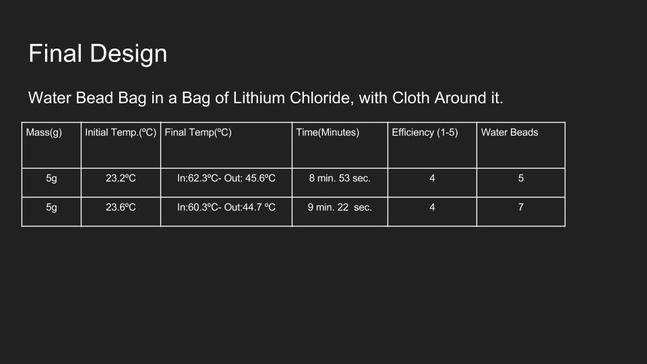
Here is the data regarding our final design. As shown by our results, the biggest problem we ran into was retaining the duration of the heat. A good hand warmer lasts for a few hours - ours lasted for just about 10 minutes. A positive for us was the heat that our hand warmer was able to attain - a comfortable 45 Celsius (or 113 Fahrenheit). The efficiency rating was just a number that we as a group came up with.
Concepts
|
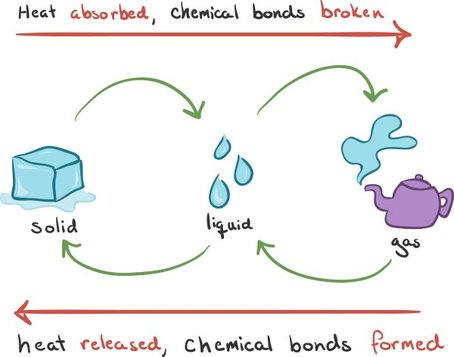
Endothermic and Exothermic Reactions
There are two main different types of reactions. Endothermic reactions are ones that absorb heat, like how in photosynthesis plants absorb heat to create glucose. Exothermic reactions are ones that release heat, like nuclear fission. To the left are examples of both endothermic and exothermic reactions with water. The arrow pointing to the left labels the endothermic reactions, while the arrow pointing to the right depicts the exothermic reaction. |
Enthalpy and Entropy
Entropy is the measure of disorder within a system. In short, it is a way of describing the amount of motion. Colder objects have a lower value of entropy, while warmer have a higher value. The same goes for gases and solids - gases have a higher value while solids have a lower value. Enthalpy is the sum of all of the energy in a system, including temperature, pressure, and volume.
Entropy is the measure of disorder within a system. In short, it is a way of describing the amount of motion. Colder objects have a lower value of entropy, while warmer have a higher value. The same goes for gases and solids - gases have a higher value while solids have a lower value. Enthalpy is the sum of all of the energy in a system, including temperature, pressure, and volume.
Reflection
This was one of the more stressful projects that I have done, mostly due to the results of the handwarmer. The most difficult aspect of our project was getting the reaction to maintain its heat for the longest time possible. We did so many trials for this sole reason. We tried using insulators, like water and charcoal. In the end, it became more difficult for us to continue to experiment with time running down on the project. Another more minor roadbump that we had was the fact that we only had one heat press - meaning that only one person could use it at a time.
With all of the loose ends, we had to wrap up the project quickly. We solidified our final design and then created our presentation. Our group worked in unison to do this, and I am happy with what our final design produced.
This project challenged our knowledge of
With all of the loose ends, we had to wrap up the project quickly. We solidified our final design and then created our presentation. Our group worked in unison to do this, and I am happy with what our final design produced.
This project challenged our knowledge of