Testing Plant Substances as Potential Medicines
Purpose
The purpose of this lab is to discover which native plants contain active ingredients that will be able to inhibit the growth of bacteria.
The purpose of this lab is to discover which native plants contain active ingredients that will be able to inhibit the growth of bacteria.
Materials
|
Plant specimen
10 mL syringe test tubes Methanol 1 mL pipet Ampicillin Inoculating Loop 60x15 Petri dish |
E. Coli JM109 (stock plate)
10 mL pipet 100 mL beakers 5mm filter paper 250 mL media bottle LB Agar LB broth base Plastic funnels |
Procedure
Part I
1. Prepare an LB broth for the E. coli. Wait 24 hours and then add a colony of the E. coli to the broth.
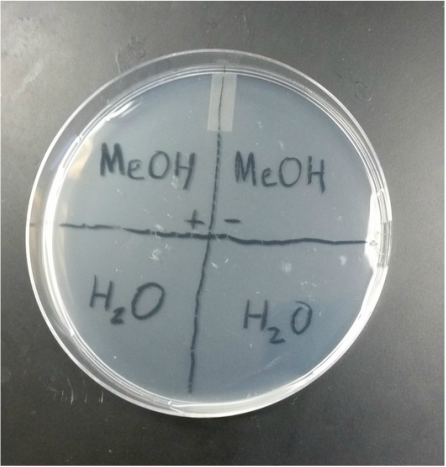
2. Get six petri dishes. Draw a "+" symbol on each plate and then divide the dish into 4 quadrants. Label the quadrants 1-4 and put your initials and the date on the plate as well.
3. Liquefy LB agar by heating it in the microwave. Pour approximately 20 mL of the agar into each petri dish. Let the agar solidify for 24 hours.
Part II
1. Grind up your plant specimen in deionized water. Filter it through filter paper. Then filter the mixture again through a syringe filter. Collect 1 mL of this mixture and put it in a labeled test tube.
2. Grind up your plant specimen in methanol. Filter it through filter paper. Then filter the mixture again through a syringe filter. Collect 1 mL of this mixture and put it in a labeled test tube. Then place the test tube in a 65 degree Celsius heat block with the cap open for 24 hours. After, put 10 mL of deionized water into the test tube.
3. Repeat steps 2 and 3 until you have 6 total samples. You will have 3 of each sample.
4. Drop 3 filter paper disks into each test tube.
5. Prepare 6 negative control disks of methanol and distilled water. (3 each)
6. Prepare 6 positive control disks of ampicillin.
7. Close tubes and store at 4 degrees Celsuis.
Part III
1. Put 1mL of E. Coli onto your petri dish and spread it around using a cleansed spreading loop. Wait for 15 minutes.
2. Place one disk in its appropriate quadrant using sterilized forceps. Make sure you have a positive and negative control disk and both sample disks.
3. Incubate the petri dishes at 37 degrees Celsius for 24 hours.
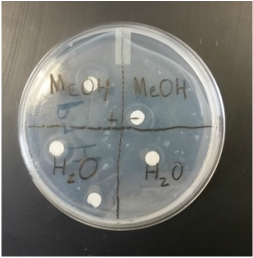
4. After 24 hours, examine the petri dishes for zones of inhibition. Photograph and record your results.
Part I
1. Prepare an LB broth for the E. coli. Wait 24 hours and then add a colony of the E. coli to the broth.
2. Get six petri dishes. Draw a "+" symbol on each plate and then divide the dish into 4 quadrants. Label the quadrants 1-4 and put your initials and the date on the plate as well.
3. Liquefy LB agar by heating it in the microwave. Pour approximately 20 mL of the agar into each petri dish. Let the agar solidify for 24 hours.
Part II
1. Grind up your plant specimen in deionized water. Filter it through filter paper. Then filter the mixture again through a syringe filter. Collect 1 mL of this mixture and put it in a labeled test tube.
2. Grind up your plant specimen in methanol. Filter it through filter paper. Then filter the mixture again through a syringe filter. Collect 1 mL of this mixture and put it in a labeled test tube. Then place the test tube in a 65 degree Celsius heat block with the cap open for 24 hours. After, put 10 mL of deionized water into the test tube.
3. Repeat steps 2 and 3 until you have 6 total samples. You will have 3 of each sample.
4. Drop 3 filter paper disks into each test tube.
5. Prepare 6 negative control disks of methanol and distilled water. (3 each)
6. Prepare 6 positive control disks of ampicillin.
7. Close tubes and store at 4 degrees Celsuis.
Part III
1. Put 1mL of E. Coli onto your petri dish and spread it around using a cleansed spreading loop. Wait for 15 minutes.
2. Place one disk in its appropriate quadrant using sterilized forceps. Make sure you have a positive and negative control disk and both sample disks.
3. Incubate the petri dishes at 37 degrees Celsius for 24 hours.
4. After 24 hours, examine the petri dishes for zones of inhibition. Photograph and record your results.
Results
Conclusion
1. What did you like/find interesting?
I found it interesting that stinging nettles could inhibit the growth of e coli. I didn't think that it was a strong enough plant to stop the bacteria because most of the leaves on the stinging nettles had been eaten away by caterpillars. I also thought it was cool how the disks created a ring where there was no bacterial growth at all.
2. How did you and your partner collaborate?
My lab partner was Poh Maga. We worked together because our sites are right next to each other and we both used stinging nettles as our sample plant. We were a good team because we were able to communicate well and finish the steps in a quick and concise manner.
3. What would you do differently next time?
I accidentally made a mistake in placing the disks in my petri dish. I accidentally put the wrong sample in the wrong quadrant. It ended up not making a difference because all the results came back positive. I also wish that I brought back other plant species to experiment on.
Think Like a Bio Technician
1. If an extract gives a negative result in the antimicrobial assay, does that mean that the extract is not an antimicrobial agent?
Most of the time, it does mean that the extract is not an antimicrobial agent. But sometimes, the extract does not diffuse properly due to differences in molar molecules.
2. In preparing the sample disk, some of the methanol extractions smell like alcohol. Why is that a problem?
Alcohol kills bacteria and this is a huge problem because it would destroy the experiment. You would not know if the extract had been altered and this would cause your results to considered false.
3. Each extract may have one or more compounds in it. What should be done to begin to identify the exact compound in an extract that is causing the antimicrobial action?
Chromatography is one method that is used to separate and identify different compounds within the extract. This separates the different compounds by their polarity. Once separted, this would allow you to test for antimicrobial action.
I found it interesting that stinging nettles could inhibit the growth of e coli. I didn't think that it was a strong enough plant to stop the bacteria because most of the leaves on the stinging nettles had been eaten away by caterpillars. I also thought it was cool how the disks created a ring where there was no bacterial growth at all.
2. How did you and your partner collaborate?
My lab partner was Poh Maga. We worked together because our sites are right next to each other and we both used stinging nettles as our sample plant. We were a good team because we were able to communicate well and finish the steps in a quick and concise manner.
3. What would you do differently next time?
I accidentally made a mistake in placing the disks in my petri dish. I accidentally put the wrong sample in the wrong quadrant. It ended up not making a difference because all the results came back positive. I also wish that I brought back other plant species to experiment on.
Think Like a Bio Technician
1. If an extract gives a negative result in the antimicrobial assay, does that mean that the extract is not an antimicrobial agent?
Most of the time, it does mean that the extract is not an antimicrobial agent. But sometimes, the extract does not diffuse properly due to differences in molar molecules.
2. In preparing the sample disk, some of the methanol extractions smell like alcohol. Why is that a problem?
Alcohol kills bacteria and this is a huge problem because it would destroy the experiment. You would not know if the extract had been altered and this would cause your results to considered false.
3. Each extract may have one or more compounds in it. What should be done to begin to identify the exact compound in an extract that is causing the antimicrobial action?
Chromatography is one method that is used to separate and identify different compounds within the extract. This separates the different compounds by their polarity. Once separted, this would allow you to test for antimicrobial action.